What is nitrate-nitrogen?
The term ‘nitrate-nitrogen’ refers to the nitrogen portion of the total nitrate in a sample. Nitrate is a common, naturally occurring chemical compound made of the elements nitrogen (N) and oxygen (O). It is an important source of the nitrogen that is essential for plants to grow. Nitrate is derived from the decay of organic material in soil and animal plant waste, and it can also be manufactured as nitrogen fertiliser.
A nitrate molecule, or ion, contains one nitrogen atom and three oxygen atoms (Figure 1).
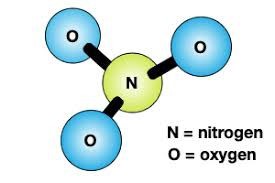
Figure 1. A nitrate molecule (ion).
The nitrogen atom is slightly smaller than the oxygen atoms, so nitrogen makes up just under one quarter of the mass of the ion – 22.6% of the mass, to be more precise. Therefore, if a sample of water contains 100 mg of nitrate, it contains 22.6 mg of nitrate nitrogen.
For environmental monitoring, scientists commonly record nitrate concentrations as nitrate-nitrogen so that they can compare the values to nitrogen in other forms that might also be present, such as ammonia nitrogen, organic nitrogen, or nitrogen gas. However, nitrate-nitrogen never occurs on its own. It is always part of the full nitrate ion.
How does nitrate enter groundwater?
Nitrate is highly soluble in water. As it forms in soil from the decay or oxidation of other nitrogen compounds, nitrate dissolves readily into the water in the soil pores. From there, plants can draw the nitrate in through their roots and use it to build proteins and amino acids that they need to grow. However, if there is more nitrate present than the plants can take up, the excess remains in the soil. Then, when it rains, that nitrate is washed, or leached, downward through the soil and into the underlying groundwater.
Therefore, nitrate contamination of groundwater occurs as a result of excess nitrate in the soil. This might be a result of excess application of fertiliser or farm effluent. It can also result from animal urine, human wastewater, or soil cultivation.
Why is nitrate a problem in groundwater?
Groundwater is commonly used as a source of drinking water in New Zealand. At high concentrations, nitrate in drinking water can pose a health risk, particularly to babies fed with formula made from the water. To prevent this risk, the Maximum Acceptable Value, or MAV, has been set at 50 mg/L for nitrate in drinking water. Note that this value refers to the concentration of the full nitrate ion; using the relationship discussed earlier, it is equivalent to a nitrate-nitrogen concentration of 11.3 mg/L. It is important to note that boiling water before drinking does not remove nitrate-nitrogen.
Some studies have suggested human health risks at concentrations much lower than the MAV. The Office of the Prime Minister's Chief Science Advisor provides a useful review of the current evidence on the health impacts of nitrates in drinking water. In addition, the Ministry of Health continues to review new evidence and research about nitrates in drinking water to understand links to adverse health. Their current advice is that nitrate-nitrogen concentrations less than 50 mg/L are safe.
Nitrate can also threaten the ecological health of surface water bodies like streams, rivers and lakes. These effects can occur at concentrations much lower than the drinking-water standard. If groundwater has high nitrate-nitrogen concentrations, any surface water fed by groundwater springs will also have high concentrations, which could cause excess plant and algae growth or be toxic to fish or aquatic invertebrates.
Why does some groundwater have very low nitrate-nitrogen concentrations?
Nitrate-nitrogen concentrations differ from one place to another for a variety of reasons, but two of the major factors are recharge source and redox conditions.
Recharge source refers to where the groundwater comes from. Some groundwater is derived primarily from rainfall that percolates through the soil. The percolating water leaches nitrate from the soil, so nitrate concentrations in the groundwater can be relatively high. In contrast, some groundwater is derived from rivers that lose some of their flow to seepage through their beds. Nitrate-nitrogen concentrations in rivers tend to be low, so groundwater recharged directly from rivers also has low concentrations.
The term redox refers to reduction and oxidation reactions – chemical reactions that remove or add oxygen to chemical compounds. Some aquifers contain organic material or vegetable matter, particularly swampy areas or that have been swamps in the geologic past. The organic material breaks down or rots over time, consuming all of the available oxygen. We call these conditions ‘reducing’.
As discussed above, the nitrate molecule contains oxygen. When nitrate is exposed to reducing conditions, the oxygen is removed and the nitrate is converted to nitrogen gas. This process is also called ‘denitrification’, and it has been shown to occur on a local scale in many parts of New Zealand. It can explain why some wells have very low nitrate concentrations when other wells nearby have much higher concentrations. However, it is unclear that denitrification plays a major role in removing nitrate from groundwater on a regional scale.
Does nitrate occur naturally in groundwater?
Nitrate is a natural chemical and it does occur naturally in groundwater, but generally at low concentrations. Estimates of natural nitrate-nitrogen concentrations in New Zealand groundwater range from as low as 0.25 mg/L (Morgenstern & Daughney, 2012) to as high as 3.5 mg/L (Daughney & Reeves, 2005). Similar ranges have been reported in the United States (Madison and Brunett, 1985; Nolan and Hitt, 2003).
How are samples tested for nitrate-nitrogen?
Water samples are collected by local authorities and sent to laboratories. Each form of nitrogen requires a different chemical test. The majority of local authorities use methods that follow the American Public Health Association (APHA) standards. Local authorities, together with the Ministry for the Environment, are currently working towards standardising sampling and testing methodologies for nutrients, including nitrogen.
In what units is nitrate-nitrogen reported?
Concentrations of nitrate nitrogen are reported on LAWA in grams of nitrogen per cubic metre (g/m3) or milligrams of nitrogen per litre (mg/L); note that these two units are equivalent and interchangeable. In some literature, concentrations may be reported as parts per billion (ppb); 1 ppb = 0.001 g/m3.
Where do I find more information?
Daughney, C., & Reeves, R. (2005). Definition of hydrochemical facies in the New Zealand National Groundwater Monitoring Programme. Journal of Hydrology New Zealand, 44(2), 105–130.
Morgenstern, U., & Daughney, C. (2012). Groundwater age for identification of baseline groundwater quality and impacts of land-use intensification – The National Groundwater Monitoring Programme of New Zealand. Journal of Hydrology, 456–457, 79–93.
Madison, R.J., and Brunett, J.O., 1985. Overview of the occurrence of nitrate in ground water in the United States. In: U.S. Geological Survey, National Water Summary 1984 – Hydrologic events, selected water-quality trends, and ground-water resources. U.S. Geological Survey Water-Supply Paper 2275, p. 93-105.
